À la une
Focus recherche
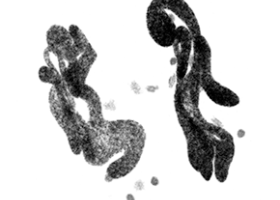
Brûler la chandelle par les deux bouts : élimination des extrémités des chromosomes chez la paramécie
Génétique, génomique
Institutionnel
GlycoMIRAI : au Japon, un laboratoire franco-japonais pour décrypter les sucres du vivant
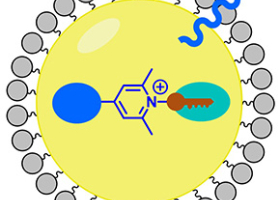
Biochimie-biologie structurale


La biologie pour tous
Des colonies de cellules aux animaux : une transition revisitée
Développement, évolution

Les cellules s'exposent au Musée d'Orsay
Au Musée d’Orsay, les scientifiques du CytoMorphoLab présentent Architecture Vivante, un spectacle où des images de cellules filmées à Grenoble se déploient à grande échelle, transformant la biologie en art.
À voir aussi
Appels à candidatures
AMI: Biologie numérique pour l’étude des systèmes dynamiques en sciences du vivant
Appels à candidatures
Appel à projets | Prix Fanny-Selena 2026 : Recherche sur l'atrésie de l'œsophage
Année thématique
Après l’Année de la Biologie, un dispositif pérenne d’échanges enseignants-scientifiques
Organisée par le CNRS et le Ministère de l’Éducation nationale, de la Jeunesse et des Sports, l’Année de la biologie vise à rapprocher les mondes de l’enseignement et de la recherche pour mettre en lumière les grandes avancées et les enjeux de la…





